NECROTIZING ENTEROCOLITIS
I. Epidemiology
A. Incidence
1. Overall national incidence ranges from 1-5% of all babies admitted to Neonatal ICUs.
2. The incidence is much higher for premature infants < 1750 grams birthweight, ranging from 5-15% of all admissions. At GHS, this figure has averaged about 5-6% for the past several years.
3. Interestingly, both antenatal steroid and postnatal steroid use have been associated with a decreased incidence of NEC. Corticosteroids are known to accelerate intestinal motility patterns, mucosal maturation and enzyme production in premature animal models.
B. Infants at Risk
NEC can occur in both premature and term infants. However, NEC predominately affects premature infants with EGA < 34 weeks and BW < 1750 grams. At risk term infants include those with cyanotic heart disease, polycythemic infants and infants suffering from birth asphyxial injury.
C. Onset
The onset of NEC has a wide range from day 1 to 3 months of age. It usually occurs between days 5 and 15 of life. 95% of these infants have been fed !!!
II. Pathogenesis
MULTIFACTORIAL. Most likely related to intermittent intestinal ischemia and/or hypoxic insult with subsequent mucosal injury. These events, in a bowel with limited motility, can cause intestinal overgrowth with pathologic bacterial organisms. The risk of NEC may be exacerbated by feedings which act to provide media for bacteria (although breast milk appears to be more protective relative to commercial formulas). Gas-producing anaerobic organisms produce the intramural gas bubbles seen on X-ray as pneumatosis intestinalis. This bowel wall damage and luminal and mural gas can cause bowel wall perforation (almost always on the anti-mesenteric side of the bowel), leading to abdominal free air.
A. Associated Factors with Intestinal Ischemia/Hypoxia
1. Asphyxia: the diving reflex (blood flow is preserved to the heart and brain at the expense of other organs).
2. Hypotension: decreased cardiac output, decreased flow to bowel.
3. Cardiac lesions: lesions associated with hypoxia and/or low cardiac output; e.g., PDA with “diastolic steal”. Decreased forward flow to bowel.
4. UAC: theoretical advantage to placement of tip below the mesenteric arteries (below L3). May decrease blood flow patterns and microemboli with
withdrawal of blood and flushing and prevent vasospasm of mesenteric arteries.
5. Exchange transfusion
6. Hyperviscosity: polycythemia, with a Hct of > 65%, may lead to hyperviscosity of the blood and subsequent perfusion problems in the bowel
7. Hypothermia: 40% reduction of GI blood flow in lambs with hypothermia.
8. Infants exposed to in utero drug usage: higher NEC rate in premies born to Mothers with cocaine exposure. This may be due to vasospasm or in utero ischemia due to the powerful vasoactive properties of cocaine. There is also evidence of an effect of cocaine on the ontogeny of vascular development.
9. Other medications: several retrospective studies have implicated indomethacin and/or ibuprofen. Other studies have implicated the underlying presence of a patent ductus arteriosus as more likely to be associated with NEC.
III. Role of Infection
When looking at the occurrence of NEC, epidemics and clustering of cases have been reported which is suggestive of an infectious process.
Blood cultures are positive in only 30-50% of infants with proven NEC.
Organisms implicated - E. coli, Klebsiella, Enterobacter, Enterococcus, Staphylococcus species, viruses and fungi.
The theory is that bacteria ferment undigested carbohydrate and this forms hydrogen gas which is seen as pneumatosis intestinalis on x-ray. Interestingly, infants fed breast milk (with its less complex nutrients and vast quantity of growth factors and IgA) or infants feed formulas supplemented with probiotics (naturally occurring enteric Lactobacilli) have a significantly less (but not nonexistent) incidence of NEC compared to formula fed infants.
IV. Role of Feedings
Feeding the premature infant is the area that generates the most discussion and controversy, probably because it is the intervention that we feel that we have the most control over. Factors in feeding that have reproducibly been shown to be associated with NEC include increased osmolality of formula, increased caloric density of formula and rapid increases in feeding volume. In the two largest retrospective and the two largest prospective studies in susceptible infants, it was shown that feeding schedules that begin in the first few days of life and advance to at least 150 cc/kg/day over the course of < 7 days result in twice the incidence of NEC as schedules that advance to full feeds over 7-14 days. Conversely, there are two smaller prospective studies that demonstrate no difference in incidence of NEC when comparing formula feeding regimens that advance to full feeds in 5 days versus 10 days. Another study found that in centers with established feeding protocols the incidence of NEC was less than in centers without such protocols. However, there were no obvious common factors noted in these “successful” protocols. Our recommendation: follow our GHS feeding protocol!
A. Whom do we feed?
B. When to feed? - waiting too long (> 2 weeks) results in microvilli atrophy and feeding difficulty. Prolonged hyperalimentation is associated with hepatotoxicity and cholestasis.
C. What to feed? Breast milk is best! But, even breast milk is not 100% protective against NEC. Otherwise, we want formulas that are isotonic (all commercially available premature formulas meet this requirement) to prevent direct mucosal injury. Lactose is hypothesized to cause some decreased intestinal motility (mucosal lactase is relatively deficient until 36 weeks post menstrual age) and perhaps undigested lactose may promote bacterial overgrowth. This hypothesis is currently being investigated (substituting other simple sugars such as maltose). Premature formula makers have also reduced the lactose content of their formulas to 40-50% of total carbohydrate. Start with un-supplemented breast milk or a 20 kcal/oz. formula, not at higher caloric densities.
D. How much to feed? - eventually need to give adequate calories (120 kcal/kg/day) for growth. For premature formulas (24 kcal/oz), this would be 150 cc/kg/day, for term formulas (20 kcal/oz), this would be 180 cc/kg/day.
E. How fast to advance? - Book, et al. found no difference in 10 cc/kg/day and 20 cc/kg/day. The NICHD study (retrospective) found that rates exceeding 10 cc/kg/day were more likely to be associated with NEC. Thus, controversy exists about which type of feeding advancement schedule is “too fast”. We suggest a conservative approach:
Early enteral feeds are o.k. It appears that hyperosmolar concerns (associated with some commercial formulas) of previous years are no longer an issue. Advancement should go slowly (at least 7 days), although there will be exceptions (e.g., IV sites run out). Some of the sickest and smallest infants may even benefit from non-nutritive feeding or gut priming (very dilute and small volume feeds, i.e., 10-20 cc/kg/day, given either continuously or every 4 - 6 hours for the first several days of life)
V. Signs and Symptoms of NEC
A. May be non-specific early:
- apnea
- temperature instability
- hypotension
- glucose intolerance
B. More specific GI signs of NEC include:
- abdominal distension
- ileus
- delayed gastric emptying (emesis, gastric residuals)
- GI bleeding
- diarrhea
- grossly abnormal stool (very foul smelling, grossly bloody, obvious pus)
- illness can range from fairly benign course to fulminant disease with shock, DIC, intestinal perforation, etc.
VI. Evaluation
A. CBC, platelets, differential
B. Blood cultures
C. Serum electrolytes
D. Blood gas
E. Other labs to consider: PT, PTT, fibrinogen, d-dimer, liver function tests, every 6-8 hours abdominal circumference
F. Abdominal x-ray: AP and a left lateral decubitus (right side up) to look for pneumatosis intestinalis and free air.50% of surgical cases which document intestinal perforation have x-rays which revealed no free air. If patient continues to deteriorate, suspect necrotic or perforated intestine.
G. Surgical consult to make them aware of patient.
REPEAT STUDIES AS CLINICALLY INDICATED. THIS MAY VARY
FROM EVERY 6-8 HOURS IN SIGNIFICANTLY ILL INFANTS TO ONCE PER DAY FOR LESS ILL INFANTS.
VII. Diagnosis
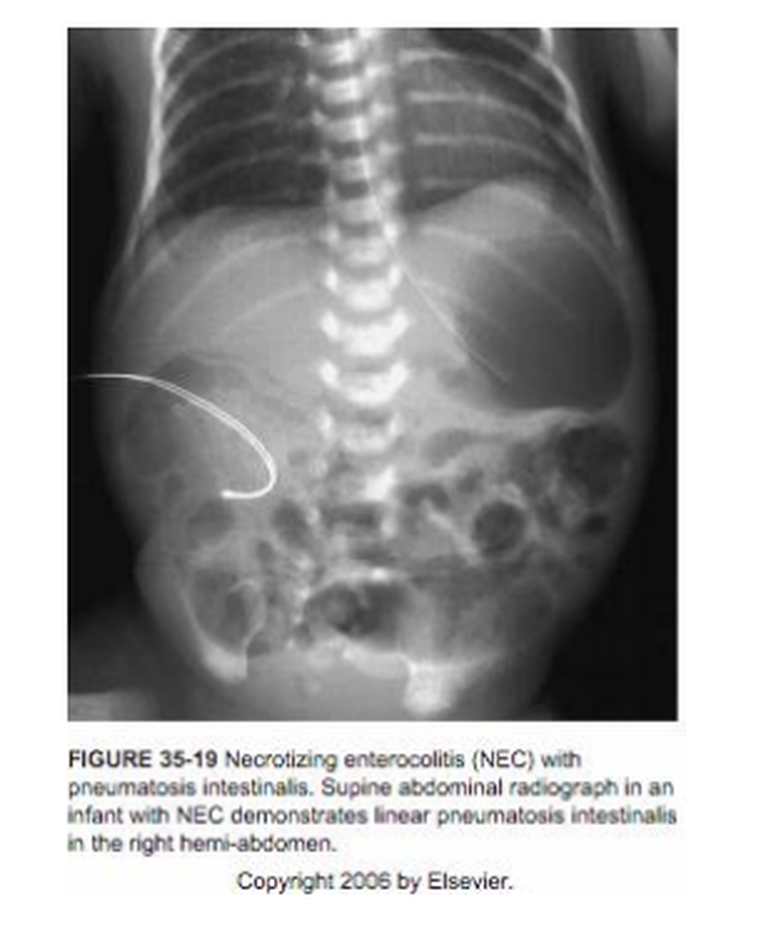
A. X-ray: pneumatosis intestinalis (right lower quadrant is most common) and/or air in the portal venous system. Free air in the peritoneum.
B. Histopathological evidence at surgery or autopsy.
VII. Treatment
A. IV antibiotics - may use ampicillin and gentamicin or substitute vancomycin for ampicillin. Add Clindamycin or Flagyl (anaerobic coverage) for severe NEC related illness or perforation.
B. NPO x 10 days for true NEC (e.g. pneumatosis intestinalis).
C. Gastric suction (low intermittent or continuous).
D. IV fluids, volume expansion, blood products as needed.
E. X-ray plus every 6-8 hours for 1st 24-48 hours and then as indicated.
F. Notify Pediatric Surgery early in the disease.
G. Notify Infection Control nurse.
References
1. Berseth, CL. Feeding strategies and necrotizing enterocolitis. Curr Opin Pediatr. 17:170-173, 2005.
2. Henry MC and Ross RL. Current issues in the management of necrotizing enterocolitis. Semin Perinatol. 28:221-233, 2004.
3. Neonatal necrotizing enterocolitis: current concepts and controversies. J Pediatr. 117(suppl):S1-S74, 1990.
4. Kleigman RM. “Neonatal necrotizing enterocolitis: bridging the basic science with the clinical disease.” J. Pediatr. 117:833-835, 1990.
5. Patole SK and de Klerk N. Impact of standardised feeding regimens on incidence of neonatal necrotising enterocolitis: a systematic review and meta-analysis of observational studies. Arch Dis Child Fetal Neonatal Ed. 90:F147-51, 2005
6. Blakely E, et. al. Postoperative Outcomes of Extremely Low Birth-Weight Infants With Necrotizing Enterocolitis or Isolated Intestinal Perforation: A Prospective Cohort Study by the NICHD Neonatal Research Network. Ann Surgery. 231:984-994, 2005.
7. Walsh MC, et al. “Necrotizing enterocolitis: treatment based on staging criteria.” Pediatr Clin of North America. 33:179-201, 1986.