NICU STUDIES, PROTOCOLS, AND PROCEDURES
The purpose of this document is to serve as a guide for residents to clinical protocols and procedures in the NICU. Materials enclosed in this document may be amended or disposed of as the clinical practice of this unit -- and Neonatology in general -- changes.
- Cranial Head Ultrasound Protocol
- Retinal Examination Protocol
- Small Baby Unit Protocols
- Audiology Exam Protocol
- Blood Transfusion Protocol and Guidelines
- Protocol for Genetic Postmortem Evaluation (Greenwood Genetics Center)
- Summary of Enteral Nutrition Protocols
- GHS Donor Breast Milk Criteria
- Vitamin Supplementation Guideline
- Steroid Therapy for Vocal Cord Edema and Bronchopulmonary Dysplasia in the NICU
- Rickets of Prematurity (Alkaline Phosphatase) Monitoring Protocol
- Prophylactic Indomethacin to Prevent IVH in Infants < 28 weeks gestational age
- Immunization Recommendations for the NICU
- American Academy of Pediatric Recommendations for Management of Infants Born to Mothers with Suspected or Presumed Group B Streptococcal Infections.
- HIV Screening recommendations
- RSV Immunoprophylaxis with Synagis®
- Neurodevelopmental Follow Up Program
- Strategies to Decrease Nosocomial Infection in the Neonatal Intensive Care
1. Guideline for CSF Reservoir Tap
2. Guidelines for the Use of Alteplase® for the Treatment of Occluded Central Line Catheters
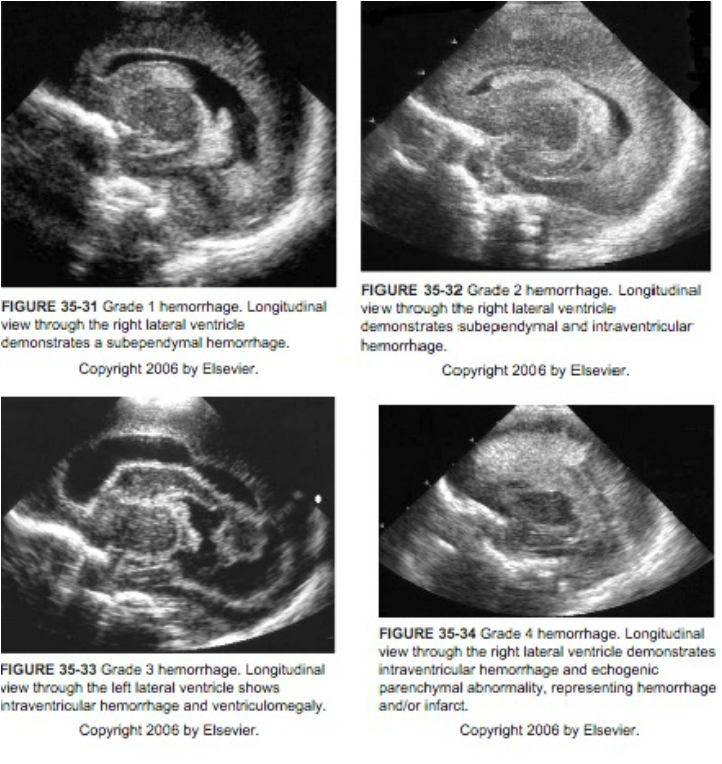
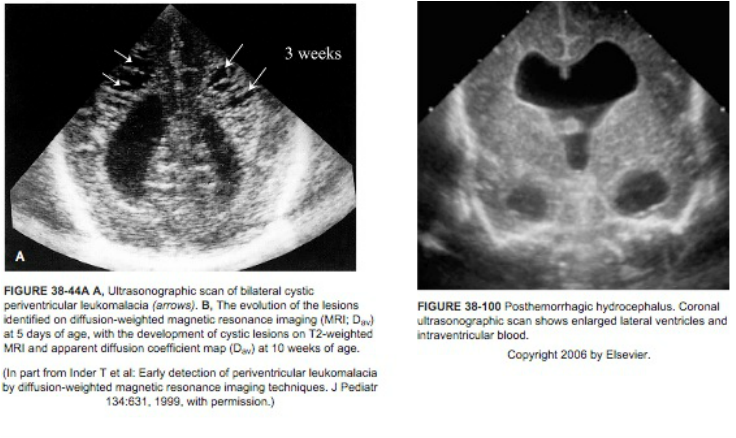
To Evaluate at-risk Infants for Intraventricular Hemorrhage (IVH) and Periventricular Leukomalacia (PVL)
- Ultrasound all infants < 31 weeks EGA (i.e., less than 31 weeks and 0 days) or < 1500 grams birth weight by 7-14 days of age (waiting until age 7-14 days insures that ~100% of cases of IVH have occurred).
- Selected infants may be screened at < 7 days at the discretion of the Attending.
- If the CUS at 7-14 days is normal the next CUS to evaluate for periventricular leukomalacia (PVL) should be obtained at 36-40 weeks postmenstrual age or prior to hospital discharge, whichever comes first. Infants scheduled for hospital discharge should have their CUS performed as an inpatient prior to discharge assuming adequate time from the previous CUS has elapsed (recommended minimum is 3 weeks).
- If the initial CUS demonstrates a Grade I, II, III, or IV hemorrhage, follow up CUS exams may be every 1-2 weeks until the lesion is stable or intervention is required.
B. Other Infants
- Routine Cranial ultrasounds of other infants should be at the discretion of the Attending Neonatologist. Typically, these exams are reserved for infants with severe pneumothorax, shock, asphyxia, meningitis, seizures, etc.
C. Procedure
- All infants will have head ultrasounds performed portable in the NICU
- Routine cranial ultrasounds should be scheduled for Monday, Wednesday, or Friday. Emergent studies can be scheduled at any time.
Ref: Practice Parameter: Neuroimaging of the Neonate. Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Ment, LR, et. al. Neurology. 58:1726-1738, 2002.
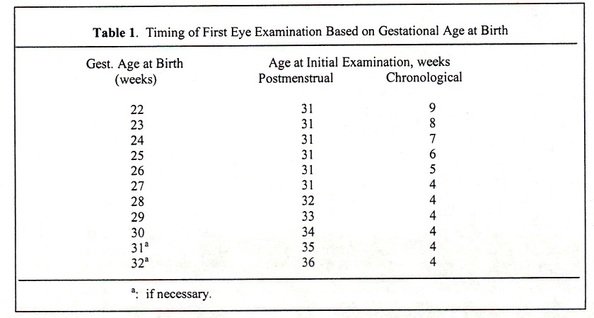
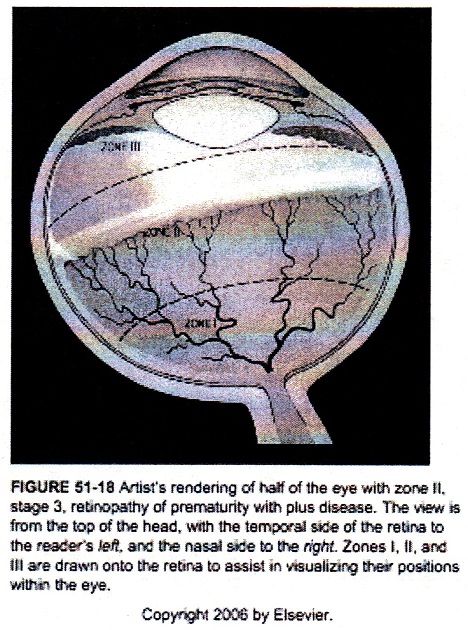
To Evaluate for the Presence of Retinopathy of Prematurity
Screening Examination of Premature Infants for Retinopathy of Prematurity . Section on Ophthamology, AAP, American Academy of Ophthamology and American Association for Pediatric Ophthamology and Strabismus. Pediatrics 2013; 131: 189-195.
Please be sure that the Retinal Exam Consult Sheet has all of the demographic information filled out, including the exact reason for the exam (ie, early onset sepsis in a 34 week premature infant > 1500 grams birth weight). Retinal exams are typically performed by Dr. Nick McLane on Tuesday. Please have eye consult request on the chart by the previous weekend.
The new recommendations for 2006 include the following.
1. all infants < 1500 grams birth weight or < 30 weeks gestational age and selected infants with a birth weight between 1500 and 2000 grams or gestational age > 30 weeks with skilled Ophthamologist using binocular indirect ophthalmoscopy. The initiation of acute-phase ROP screening should be based on infant’s age (post menstrual age correlating better with onset of severe ROP than postnatal age – see Table 1 below).
2. Follow up examinations should be based on the consulting Ophthamologist recommendations. A suggested schedule is shown in Table 2.
3. Finally, communication with parents is very important. Parents should be aware of the exam and should be informed if their infant is discovered to have ROP. These conversations should be documented in the chart. Parents and primary care physicians must be made aware of any infant requiring follow up examinations after transfer or discharge to home. It is recommended that all follow up exams be scheduled with the consulting Ophthamologist prior to infant transfer or discharge to home.
Suggested criteria for 31-37 week gestational age premature infants who may benefit from ROP screening:
a. > 3 days on the ventilator with a significant oxygen requirement
b. > 7 days of nasal CPAP or nasal cannula oxygen
c. any infant going home on medications for “chronic lung disease”
d. any infant with confirmed or highly suspicious sepsis (i.e., NEC)
e. any infant born with fetal hydrops
f. any infant born with congenital viral infections
g. any infant who is both SGA and < 37 weeks EGA
h. any infant requiring > 2 transfusions for anemia (hemolytic or non-hemolytic)
(In 1994, the AAP recommended screening tests for all babies prior to discharge from the hospital). The following risk factors are indicators for newborn hearing screening as listed in Section III E of “ Principles & Guidelines for Early Hearing Detection and Intervention Programs, Joint Committee on Infant Hearing 2000 Position Statement,” Audiology Today, August 2000, Special Issue.
Birth through age 28 days:
An illness or condition requiring admission of 48 hours or more to a NICU
Stigmata or other findings associated with a syndrome known to include a sensorineural and/or conductive hearing loss
Family history of permanent childhood sensorineural hearing loss
Craniofacial anomalies, including those with morphological abnormalities of the pinna and ear canal
In-utero infection with cytomegalovirus, herpes, toxoplasmosis, or rubella.
Age 29 days through 2 years of age:
Parental or caregiver concern regarding speech, hearing, language, and/or developmental delay
Family history of permanent childhood hearing loss
Stigmata or other findings associated with a syndrome known to include a sensorineural and/or conductive hearing loss or Eustachian tube dysfunction
Postnatal infections associated with sensorineural hearing loss including bacterial meningitis
In-utero infection with cytomegalovirus, herpes, toxoplasmosis, or rubella
Neonatal indicators-specifically hyperbilirubinemia at a serum level requiring exchange transfusion, PPHN associated with mechanical ventilation, and conditions requiring the use of ECMO
Syndromes associated with progressive hearing loss such as neurofibromatosis, osteopetrosis, and Usher’s syndrome
Neurodegenerative disorders, such as Hunter syndrome, or sensory motor neuropathies, such as Friedreich’s ataxia and Charcot-Marie-Tooth syndrome
Head trauma
Recurrent or persistent otitis media with effusion for at least 3 months.
When to screen infants in the NICU setting
1. > 36 weeks post menstrual age, or > 1500 grams
2. Within 24 hours after discontinuation of potentially ototoxic antibiotic therapy
3. Generally should be ordered at least 1 week prior to discharge.
4. Currently performed by the Neonatal Audiology Screening Team at the Greenville Hospital System. They will document in the patient’s chart whether or not infant passes or fails the screen, and will arrange any outpatient rescreening, if necessary.
Transfusion Triggers:
Hematocrit < 30% (recommended) and Hematocrit < 40% (optional)
1. Severe Pulmonary Disease requiring:
a. FIO2 > 35% and/or
b. CPAP > 6 cm and/or
c. Mechanical ventilation with MAP > 6 cm
2. Congestive heart failure with anemia contributing to the problem
Hematocrit < 25% (recommended) and Hematocrit < 30% (optional)
1. Pulmonary Disease requiring:
a. Nasal cannula oxygen > 200 mL/min
b. FIO2 < 35% and/or
c. CPAP > 6 cm
d. Mechanical ventilation with MAP < 6 cm
2. Significant apnea and bradycardia defined as
a. > 9 stimulation-requiring episodes in 12 hours
b. 2 episodes in 24 hours requiring BMV intervention
3. Persistent tachycardia (HR > 180) and tachypnea (RR > 80) without other explanation
4. Unacceptable weight gain despite adequate caloric intake
5. Pre-operative patients for major surgical procedures (in consultation with Pediatric Surgery)
Miscellaneous:
1. Transfuse for Hct < 20% with a reticulocyte count < 1% (required)
2. Shock not responsive to initial crystalloid therapy
3. Do not transfuse for “blood out”
4. These guidelines do not apply to clinical situations related to exchange transfusion due to hemolytic anemia or hydrops fetalis.
TRANSFUSION AND FEEDING PROTOCOL
After sifting through the available literature there is still some confusion as to the exact relationship between transfusions and the development of NEC. The following statements seem to be agreed to by a number of the articles.
1. Anemia is associated with an increased risk of NEC. The more anemia, the greater the risk.
2. There is a temporal relationship between blood transfusions and development of NEC.
3. Greatest risk for NEC following transfusions is 24-48 hours after the transfusion. Relation of transfusion and NEC seems to significantly decrease after 48 hours post transfusion.
4. Some authors feel that transfusing anemic infants puts these infants at greater risk of developing NEC.
5. Holding feedings “around” transfusion may be beneficial to infants and decrease the risk of developing transfusion related NEC.
6. Transfusions lead to blunted intestinal hyperemic response in infants to feedings immediately following the transfusion. How long there is this blunted response is unknown.
7. Adults may have decreased mesenteric perfusion following transfusion for at least 6 hours.
8. There is literature that transfusion related NEC is seen in infants of all postnatal ages and smaller infants seems to be at highest risk.
9. Transfusing blood greater than 12 days old may carry and increased risk of transfusion related NEC but this opinion is not shared by all authors.
10. Our Blood Bank will begin with units that have at least 21 days left on them (Blood is 1-21 days old to start) and then stick with that unit until it is gone or it expires at 42 days. This is consistent with philosophy of minimizing donor exposure.
Considering the above the following is a draft of how we should handle development of anemia, transfusions and feeding.
A. We should minimize amount and frequency of phlebotomy. Begin iron at appropriate time. Consider twice a day iron for growing premies. The changes in glucose draws and ionized calciums are an initial attempt at this. We should stop doing what I call “knee jerk” labs that have little clinical relevance.
B. Feedings should be held around all transfusions. The question is how.
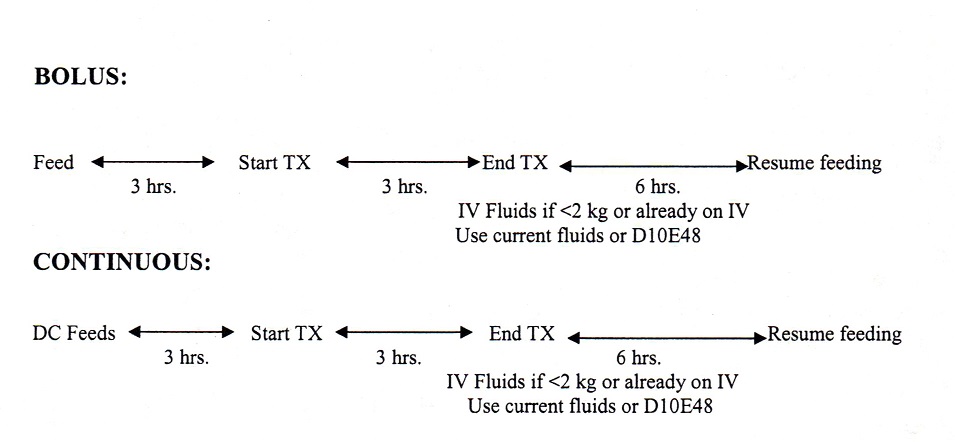
1. For bolus feeding the transfusion should begin no sooner than three hours after the last feeding. Transfusion should be given over 3 hours (unless being given for volume replacement). The next two feedings should be held after the transfusion (this would take us out to about 6 hours post transfusion. For the 6 hours of NPO after the transfusion, if the infant is already on IV fluids, run current IV fluid at daily TF amount, then resume feeding and adjust IV rate appropriately. If not on IV fluid and infant is <2 kg., run some D10E48 for the 6 hours then resume feeds and DC IV fluids.
2. For continuous feedings stop the feeding 3 hours before transfusion begins. Transfuse the blood over 3 hours (unless being given for volume replacement). Wait 6 hours post transfusion to begin continuous feedings. For the 6 hours of NPO after the transfusion, if the infant is already on IV fluids, run current IV fluid at daily TF amount, then resume feeding and adjust IV rate appropriately. If not on IV fluid and infant is <2 kg., run some D10E48 for the 6 hours then resume feeds and DC IV fluids.
For all miscarriages, stillbirths and infant deaths with any structural birth defects or suspected genetic or metabolic diseases, you are strongly urged to contact Dr. Curtis Rogers, Dr. David Everman of Genetics or the Greenwood Geneticist on call:
1. Drs. Everman/Rogers: office 250-7944, home (Rogers) 879-2390, beeper 1544.
2. Greenwood Genetics Center: (864) 941-8100.
Highly Recommended in All Cases (either premortem or within 2-4 hours of death)
1. Genetics Consultation - see above
2. Full Body X-ray (“the babygram”): particularly any infant with a skeletal dysplasia or other skeletal abnormality.
3. Photographs if appropriate (a digital camera and printer is available in the NICU and in L&D).
Recommended in Most Cases
4. Karyotype (2-4cc blood in a Sodium Heparin Green Top Tube to be sent for high resolution chromosomal analysis at the Greenwood Genetic Center Cytogenetics Lab. Can also be used for plasma amino acid analysis.)
5. DNA Analysis: skin biopsy (1 cm) transported in Hank’s Media to the Greenwood Genetic Center Molecular Genetics Lab for fibroblast growth, immortalization and archiving, DNA analysis and Gene Analysis. 3-4 cc of blood in a yellow top tube (room temperature) can be sent for molecular DNA analysis
6. Metabolic Studies: Urine (5-10 cc) frozen and sent to Greenwood Genetic Center Mass Spectrometry Lab for Organic and Amino Acid analysis.
7. Can also request other samples for fatty acid deficiencies, carbohydrate deficiencies and specific enzymatic deficiencies after consultation with a Genetic representative (usually 3-4 cc in a red top tube kept at room temperature).
Recommended Following Parental Consultation and Permission
8. Genetic autopsy at Greenwood Genetic Center Anatomic Pathology Lab can only be performed in cases in which anatomical, chromosomal, or metabolic abnormalities are known or suspected. Check with the GCC before offering their autopsy services to parents. All other autopsies must be done here
All fluids and tissues can be sent through the NICU Lab. Autopsy referral is through the Greenville Hospital System Pathology Department/Greenville County Coroner’s Office.
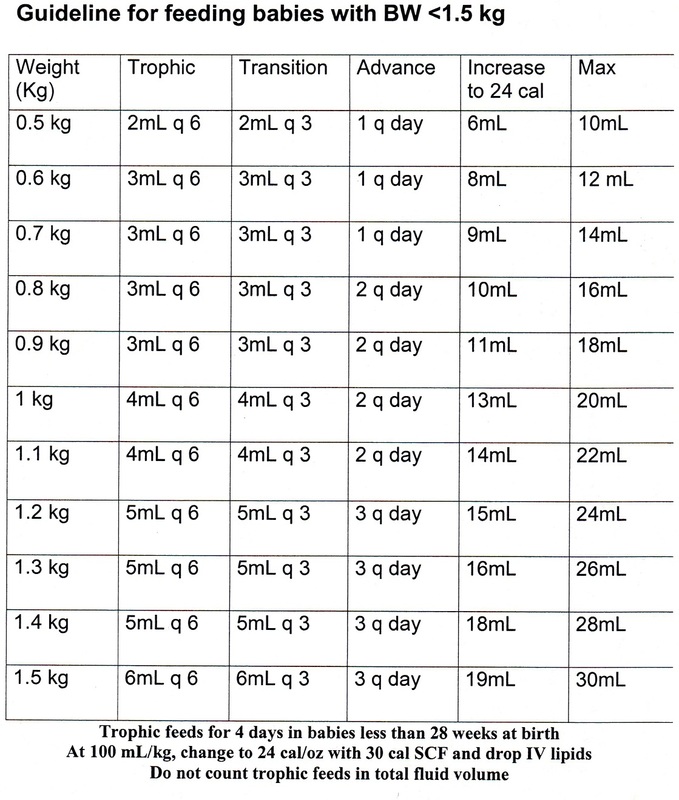
FEEDING GUIDELINE SUMMARY (for infants less than 1500 gm):
Trophic Feeds : in q 6hr increments, over 1 hour (15ml/kg/day)
Caloric Feeds : in q 3hr increments, over 1 hour until reach full volume feeds
All feeds will be compressed over 1 hr, even with CPAP and Ventilator support, unless there is feeding intolerance.
Always check NG/OG placement per policy
Always include all feedings in total daily fluid volume
Change to 24 cal/oz when at 100ml/kg/day
RESIDUALS (for all babies):
DO NOT check residuals unless there are signs of feeding intolerance such as abdominal distention, emesis
If there are residuals during caloric feedings-
<50% of feeding, re-feed residual, subtract volume from feeding, and continue advance
>50% of feeding, re-feed residual, subtract volume from feeding, and hold advance until residuals are <50%
ALWAYS notify NNP/Resident :
- if residuals >50% 2 feedings in a row
-green residuals after 1 week of age
-abdominal distention/discoloration/bloody stool
-emesis
DO NOT make an infant NPO without an order
With signed consent:
· Infant born < 32 weeks gestation
· All infants < 1500 grams until they reach 34 weeks (longer if they meet criteria for extended use)
- Post NEC infants
- GI anomalies/surgery
- Feeding intolerance
- Malabsorption syndromes
- Short-gut syndrome
- Cardiac
- Metabolic errors
High Calorie Donor Milk Guidelines:
· Infants < 1 kg on mostly donor milk and poor growth
· Infants < 1 kg unable to tolerate HMF
How to mix:
· 26 cal/oz : Use 22 cal/oz donor milk and add HMF 1 packet per 25 mL donor milk
· 28 cal/oz: Use 24 cal/oz donor milk and add HMF 1 packet per 25 mL donor milk
9. Vitamin Supplementation Guideline
Our steroid protocol for alleviation of the inflammatory symptoms associated with developing or established lung disease in the NICU are changing in response to recent neonatal literature calling into question the short and long-term benefits of postnatal corticosteroid exposure. Significant side effects of systemic steroid therapy in the preterm neonate occur both acutely (hypertension, hyperglycemia,failure to gain weight, cardiac ventriculomegaly, GI tract bleeding or perforation,immunosuppression, adrenal suppression) and long-term ( increased risk of severe neuromotor developmental delay and head growth impairment). Nevertheless, there are acute benefits in reduction of ventilatory support and supplemental oxygen exposure.
1. Barrington KJ. “Postnatal steroids and neurodevelopmental outcomes: A problem in the making.” Pediatr. 107:425-426, 2001.
2. Thebaud B, Lacaze-Masmonteil T, Watterburg K. “Postnatal glucocorticoids in very preterm infants: “the good, the bad, and the ugly.” Pediatr. 107:413-415, 2001.
3. Committee on the Fetus and Newborn. “Postnatal corticosteroids to treat or prevent chronic lung disease in preterm infants.” Pediatr 109:330-338, 2002.
STEROID PROTOCOLS FOR VOCAL CORD EDEMA AND “BRONCHOPULMONARY DYSPLASIA”
Dexamethasone 0.25-0.50 mg/kg/dose IV every 4-6 hours for 4-6 total doses. The objective should be to extubate the infant after the first 1-2 doses of therapy in order to allow further steroid benefit after the ETT is removed.
2. “Bronchopulmonary Dysplasia” (intubated)
DART protocol: Use dexamethasone (Decadron) IV dart taper in Epic. 0.075 mg q12 x 6 doses, then 0.05mg/kg q12 x 6 doses, then 0.025mg/kg q12 x 4 doses, then 0.01 mg/kg q12 x 4 doses
Pulse Dexamethasone: 0.10-0.30 mg/kg/day (divided BID) X 3 days, then wait 7-10 days before considering another pulse dexamethasone administration. This therapy generally should only be reserved for intubated patients. The objective of this therapy is to allow significant weaning from high oxygen levels (FIO2 > 0.8-1.00) and/or high ventilatory pressures. Benefits should be seen within 24-48 hours after institution of therapy.
3. “Bronchopulmonary Dysplasia Exacerbation” (extubated)
There is no specific protocol because there is no data. The decision to use steroids is left to the discretion of the patient’s management team and its consultants. Parenteral, enteral, and inhaled steroids are all options.
At risk infants for Rickets of Prematurity:
1. Any infant < 1000 grams birth weight
2. Any infant on prolonged hyperalimentation (ie, infants in whom HAL provides the majority of calories for > 30 days; either intermittently or cumulatively)
3. Any infant with major gastrointestinal anomalies (ie, short bowel disease, prolonged feeding intolerance, major gastroschisis or omphalocele, hepatic failure, etc.)
Obtain serum Alkaline Phosphatase concentration at 6 weeks after birth. Levels should be less than 5-6 times the upper limits of normal for adults, ie, < 600 IU/L). If the infant has a level > 600 IU/L, consider starting ergocalciferol supplementation 2 weeks along with radiographic studies depending on the clinical circumstances. Careful calculation of calcium and phosphorus intake may be required along with further metabolic workup of persistent unexplained elevated AlkPhos levels in the face of adequate caloric, mineral, and vitamin intake.
Reference: Nutritional Needs of the Preterm Infant, Tsang RC, et al, Editors, 1993.
(Indomethacin use to close symptomatic PDAs in other infants is discussed in Chapter 7)
Cochrane Collaborative meta-analysis of well designed clinical trials using prophylactic Indomethacin therapy to prevent severe IVH demonstrates that prophylactically administered Indomethacin begun before 12 hours of age in preterm infants, (generally < 1500 gms birthweight) decreases the incidence of severe IVH in this population. The presumed mechanism of action is via stabilization of cerebral blood flow and/or closure of an open PDA.
Other retrospective studies have raised cautionary flags about this regimen as there has been no improvement in long-term neurodevelopmental outcomes in sometreated groups versus controls despite lower rates of severe IVH in the treated groups. Another concern regarding prophylactic Indocin therapy appears to be an association with an increased incidence of isolated intestinal perforation, presumably be decreasing regional blood flow to the distal ileum. Several covariables may contribute to this latter risk: the usual practice of bolus therapy (< 5-20 minutes), the usual presence of a “high” UAC (e.g. above the branch of the superior mesenteric artery off of the aorta at T12), the use of higher doses (0.2-0.25 mg/kg) of Indocin, and the use of antenatal and early postnatal (< 7 days of age) steroid therapy.
Our practice has had a lower incidence of severe IVH versus the Vermont-Oxford database for several years using prophylactic Indocin therapy in infants < 28 weeks gestational age (~ 10% versus 15%). PDA ligation in our practice is less than 50% of that seen in the VON database.
Using appropriate precautions, it seems rational to try to continue this practice of reducing pathological IVH in our lowest birth weight population. In addition, we hope to gain the additional benefit of decreasing the severity of PDA effects by adopting some of the most successful prophylactic Indocin protocols. Thus, our protocol will try to take advantage of lower and slower doses of Indocin in appropriate high-risk infants.
General Indications:
1. < 28 weeks estimated gestational age (90% will have a ductus at 12-24 hours of life)
2. Not severely asphyxiated, platelet count > 50,000 and mild-to-moderate bilirubin levels
3. No major electrolyte or renal disorder
4. Attending approval
Dose : 0.1 mg/kg IV over 60 to 120 minutes at 6 - 12 hours postnatal age and repeated q 24 hrs x 2 additional doses. ECHO: there is a risk of closing a ductal dependent lesion in these babies (PGE1 would be the therapy of choice if one believes this has happened). A pretreatment Echocardiogram may be considered in the most high-risk premature infants but is not necessary. An ECHO should be performed following prophylactic indomethacin therapy to evaluate for closure of the PDA.
Reference: Ment, et al. “Low-dose Indomethacin and Prevention of Intraventricular Hemorrhage: A Multicenter Randomized Trial.” Pediatr. 93: 543-550, 1994 and the Cochrane Collaborative Database (www.nichd.nih.gov/cochraneneonatal, 2002).
Consider the risk / benefit ration of initiating or continuing Indocin therapy in ALL PATIENTS .
1. Grade 3 or 4 IVH
2. Excessive bleeding at venipuncture sites, capillary sticks, platelet count < 50,000, etc.
3. Pulmonary or GI hemorrhage
4. Clinical or X-ray evidence of NEC or perforation
5. Elevated serum creatinine (> 1.5) or oliguria, elevated serum K+ (> 6.0)
6. Elevated serum bilirubin
7. Severe birth asphyxia
8. Hepatic dysfunction
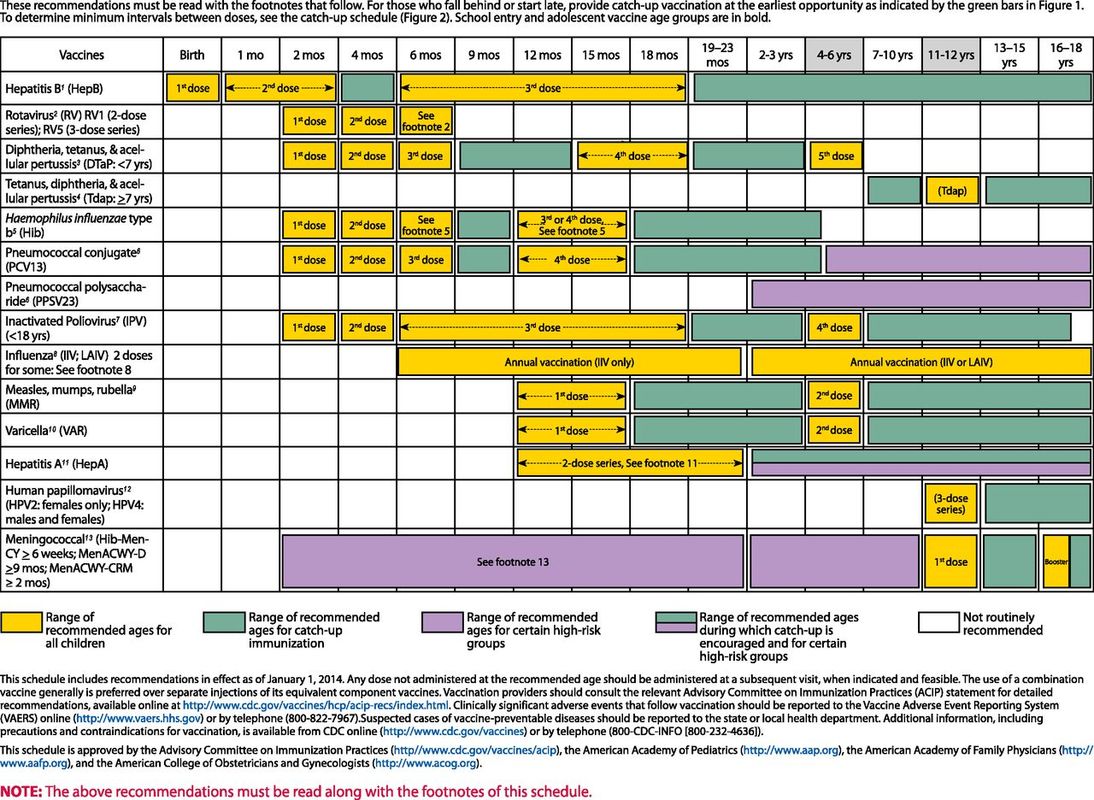
Recommended childhood immunization schedules are listed on the following pages as promoted by the Advisory Committee on Immunization Practices, the American Academy of Pediatrics and the American Academy of Family Physicians (Pediatrics 2013; 131:2 397-398). Specific discussion of each of the routine immunizations given to patients in the Neonatal Intensive Care setting are discussed in the paragraphs below.
REMEMBER TO USE COMMON SENSE BEFORE GIVING VACCINES! If a neonate is critically ill or severely immunocompromised (with acute sepsis, HIV infection, unexplained neutropenia, recent steroid exposure, etc.), it may be prudent to wait or alter vaccine schedule. Check with your Attending .
A. COMBINATION VACCINES (PediarixTM GlaxoSmithKline):
PediarixTM (GlaxoSmithKline) is a combination immunization created from tetanus toxoid, diptheria toxoids, acellular pertussis proteins (there are 3), hepatitis B surface antigen and inactivated polio vaccine. It is meant to be given as a single 0.5 mL intramuscular injection at 2, 4, and 6 months of chronological age in all infants, no matter how premature they may be. No combination vaccine should be used on infants < 6 weeks of age. The DTaP components are the same as those found in Infanrix® and the hepatitis B surface antigen is the same as that found in Engerix-B®.
Side effects include redness and swelling at the site of immunization, low grade fever and irritability in up to 40-50 percent of the infants. With the acellular pertussis component, the frequency of severe side effects is much less than those associated with the whole cell pertussis vaccines. Severe adverse reactions to any vaccine should always be reported to the Vaccine Adverse Event Reporting System (VAERS, www.vaers.org or by calling 1-800-822-7967).
Procedure: for infants who are still in the NICU at 2, 4, or 6 months of age, they are eligible for a PediarixTM immunization. After consulting with the attending physician and before placing an order for this immunization in the chart, make sure that a signed consent form for vaccination (available at the front desk) has been read and signed by the parent and is on the chart. Each dose is 0.5 mL given intramuscularly in the upper lateroanterior quadrant of the thigh. Prophylactic acetaminophen may be ordered for the subsequent 24 hours. It is critical when giving PediarixTM, not to order separate doses of DTaP, IPV, or Hepatitis B vaccine as PediarixTM contains all 3 components .
B. INACTIVATED POLIO VACCINE (now part of the PediarixTM vaccine)
Polio vaccine is given to term and preterm infants using the same dosing schedule as PediarixTM. It is critically important to remember that inactivated polio vaccine is the only choice in the NICU setting. Oral polio vaccine (OPV) is an attenuated live vaccine that can be shed for several months in an infant’s stool. Surrounding significantly immunocompromised neonates may be at risk for full-blown polio infection if accidentally inoculated.
Procedure: for infants who are still in the NICU at 2, 4, or 6 months of age, they are eligible for a IPV immunization. After consulting with the attending physician and before placing an order for this immunization in the chart, make sure that a signed consent form for vaccination (available at the front desk) has been read and signed by the parent and is on the chart. Each dose is 0.5 mL given subcutaneously in the upper lateral anterior quadrant of the thigh (at a separate site from other immunizations that are being given concurrently). Side effects are less frequent but similar to DTP immunizations and acetaminophen therapy for 24 hours following immunization is appropriate.
C. HEPATITIS B VACCINES
Hepatitis B remains a serious disease in the United States. Approximately 5% of the population has been infected and 0.2% to 0.9% are chronically infected. Of concern for infants, the rate of vertical transmission of HepB from infected mothers (HBeAg-positive) to infants is 70%-90%. Unfortunately the conversion rate to chronic infection in neonates is 80%-95% vs 1%-10% in older children and adults. The incidence of chronic infection progressing to end stage liver disease or hepatocellular carcinoma is 20%-50% in chronically infected neonates.
Thus, it is currently recommended that all pregnant women be screened for Hepatitis B and that all newborns be immunized against Hepatitis B. In addition, all infants born to mothers with active or chronic Hepatitis should receive Hepatitis B IVIG ( HBIG - 0.5 mL IM) in the first 12 hours following delivery.
The currently recommended dosing schedule for infants of Hepatitis B vaccine is prior to discharge from the hospital, at 1-2 months of age and again at 6 months of age. There are two Preservative-free vaccines on the market in the US; the Recombivax HB (Merck) and the Engerix-B® (GlaxoSmithKline). Each is dosed at 0.5 mL intramuscularly. The Greenville Hospital System currently carries the Engerix-B® vaccine. We only use monovalent Hepatitis B vaccines for the birth dosing!
Low-risk infant (HBsAg-negative mother) 10 ug (0.5 mL)
High-risk infant (HBsAg-positive mother, or unknown status) 10 ug (0.5 mL) plus
HBIG also recommended 0.5 mL IM
Infants born to mothers with an unknown HBSAg status should be given HepB vaccine within the first 12 hours after birth while awaiting determination of mothers’ serum HBSAg status. If the Mom is found to be HBSAg+, then HBIG should be given at a separate physical site from the HepB vaccine as soon as possible. Otherwise, all infants > 2 kilograms birth weight in the NICU should receive the HepB vaccine at the earliest convenient moment provided that (1) he/she is clinically stable, (2) has met attending approval and (3) has documented parental permission documented on the chart. Additional doses should be scheduled at 1-2 months and 6-18 months of chronological age (can be given on separate days from the Pediarix or Prevnar immunizations).
Preterm infant immunization with Hepatitis B vaccine remains controversial primarily because the seropositive conversion rate of preterm infants following 3 doses of Hepatitis B vaccine remains less than comparative rates from term babies (~80% vs 100%). The current Red Book recommendations (from the Report of the Committee on Infectious Diseases) for Hepatitis B immunization states that preterm infants (< 2 kg at birth) should have immunization for HepB delayed until the infant is 30 days old. However, we think that a critically opportune time for immunization is missed if an infant is discharged from our Neonatal Intensive Care Unit without having received HepB. Thus, we recommend that all infants be immunized with HepB prior to discharge from the hospital irregardless of discharge weight:
1. Infants > 2000 gms birthweight: when clinically stable (usually the 1st 24 hours of life)
2. Infants < 2000 grams birthweight: at 30 days of age or prior to discharge, whichever comes first.
PrevnarTM, a hepatavalent pneumococcal vaccine (PCV7) manufactured by Wyeth is recommended for all infants at 2-23 months of age (at 2, 4 and 6 months of age and again at 12-18 months of age) and for certain children 24-59 months of age. In addition, a 23 valent polysaccharide vaccine (PPV) is recommended is certain high risk groups of children (but it is not recommended for infants). It is administered as a 0.5 mL intramuscular injection at a site different from other IM vaccines administered on the same day. All premature infants should also be immunized with PrevnarTM at 6-8 weeks of age, regardless of their original birth weight.
E. INFLUENZA VACCINES
Generally, these are multivalent inactivated vaccines containing 3 virus strains (usually 2 type A and 1 type B). It is currently recommended that all infants and children 6-59 months be considered for inactivated influenza vaccination. The dose is 0.25 mL administered intramuscularly. Optimal immunogenicity requires two separate doses administered > 4 weeks apart for recipients of the first time trivalent immunization. In the NICU, the ideal candidate is the infant with bronchopulmonary dysplasia now 6 months old chronologically. Attenuated live virus vaccines are available for older children.
F. Rotavirus
Because the only currently available vaccine for rotavirus contains live attenuated human virus, it is not routinely used in the NICU.
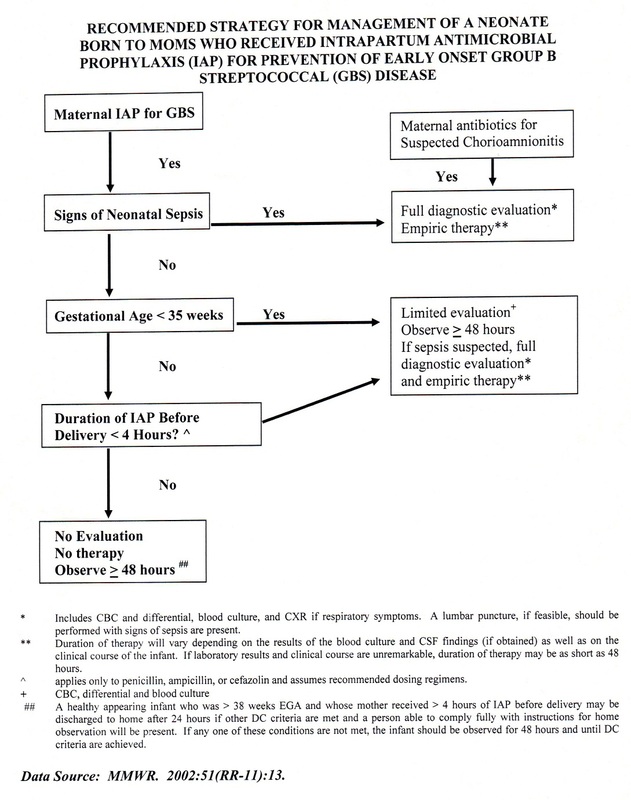
14. AAP Recommendations for Management of Infants Born to Mothers with Suspected or Presumed Group B Streptococcal Infections.
The current recommendations of the American Academy of Pediatrics and the authors of the 2012 Red Book are that documentation, HIV education and routine consent and testing for HIV be performed on all pregnant mothers. It is recommended that all infants born to mothers whose HIV status is unknown be tested for HIV following maternal education and consent approval. The Greenville Hospital System strongly advocates prenatal HIV testing, counseling and treatment as necessary. The routine testing of infants for HIV is still being debated. Nevertheless, it is clear that early treatment of HIV-exposed infants in the first few hours of life dramatically decreases their risk of HIV conversion. We encourage diligent review of maternal charts for HIV status. Mothers without prenatal HIV testing or who refuse HIV screening should have an order placed in the infant’s chart for a rapid HIV screen that may be ordered STAT from the lab (4 hour turnaround, 0.9 mL of blood in a peach top tube). A standard request for Stat Rapid HIV Testing is available at the NICU front desk.
When testing a mother, a consent form must be obtained for STAT HIV status testing. When testing the infant, the consent form is NOT required.
Mothers who refuse consent or whose HIV medical history is incomplete should be discussed with an attending neonatologist in the first day of life . Initiation of zidovudine therapy more than 48 hours after birth in an infant born to an HIV-positive mother is not likely to be as efficacious as therapy started shortly after birth.
The following therapies are currently recommended for HIV exposed infants and mothers:
Infant dosing of AZT (zidovudine):
1. 2 mg/kg/dose po Q 6 hours (for infants > 36 weeks at birth) for the first 6 weeks of life
2. 2 mg/kg/dose po or IV Q 12 hours (for infants 30-36 weeks at birth) for the first 2 weeks of life, and then Q 8 hours for the next 4 weeks
3. 2 mg/kg/dose po or IV Q 12 hours (for infants <30 weeks at birth) for the first 4 weeks of life, and then Q 8 hours for the next 2 weeks
Finally, please notify Dr. Robin LaCroix or Dr. Sue Jue of any infant of an HIV+ mother or an infant who has a positive HIV screen
RSV, a large, enveloped RNA virus, is a member of the paramyxovirus family with two major strains; A and B. Humans are the only source of infection. The virus is transmitted by close contact or secretions and it can persist on environmental surfaces for many hours. RSV occurs in annual epidemics, usually in the spring and winter. The period of viral shedding is 3 to 8 days, but may be longer in young infants (3 to 4 weeks). The incubation period prior to infection ranges from 2 to 8 days. Reinfection throughout life is common.
Estimates of hospitalization for RSV infections range from 40,000 to 90,000 annually in the United States. 2000 to 4000 deaths are reported annually, usually infants and at-risk children and adults. It is estimated that up to 5-10% of all premature infants < 32 weeks gestation at birth need rehospitalization secondary to RSV infections in the first year of life. Because no vaccine to date has been developed, the American Academy of Pediatrics and the 2012 Red Book Committee have recommended immunoprophylaxis with an IM monoclonal antibody preparation known as (SYNAGISTM) for the most at-risk infants (see trial results in the September issue of Pediatrics 102(3): 531-537, 1998).
The AAP updated their recommendation in 2009 particularly with regard to infants 32 weeks, 0 days through 34 weeks, 6 days with either daycare attendance, or a sibling less than 5 years of age. The following summarizes the changes :
SUMMARY OF MAJOR CHANGES TO THE RECOMMENDATIONS FROM THE 2009 AAP STATEMENT
(Currently up to date as of June 2018)
1 In the first year of life, palivizumab prophylaxis is recommended for infants born before 29 weeks, 0 days’ gestation.
2 Palivizumab prophylaxis is not recommended for otherwise healthy infants born at or after 29 weeks, 0 days’ gestation.
3 In the first year of life, palivizumab prophylaxis is recommended for preterm infants with CLD of prematurity, defined as birth at <32 weeks, 0 days’ gestation and a requirement for >21% oxygen for at least 28 days after birth.
4 Clinicians may administer palivizumab prophylaxis in the first year of life to certain infants with hemodynamically significant heart disease.
5 Clinicians may administer up to a maximum of 5 monthly doses of palivizumab (15 mg/kg per dose) during the RSV season to infants who qualify for prophylaxis in the first year of life. Qualifying infants born during the RSV season may require fewer doses. For example, infants born in January would receive their last dose in March.
6 Palivizumab prophylaxis is not recommended in the second year of life except for children who required at least 28 days of supplemental oxygen after birth and who continue to require medical intervention (supplemental oxygen, chronic corticosteroid, or diuretic therapy).
7 Monthly prophylaxis should be discontinued in any child who experiences a breakthrough RSV hospitalization.
8 Children with pulmonary abnormality or neuromuscular disease that impairs the ability to clear secretions from the upper airways may be considered for prophylaxis in the first year of life.
9 Children younger than 24 months who will be profoundly immunocompromised during the RSV season may be considered for prophylaxis.
10 Insufficient data are available to recommend palivizumab prophylaxis for children with cystic fibrosis or Down syndrome.
11 The burden of RSV disease and costs associated with transport from remote locations may result in a broader use of palivizumab for RSV prevention in Alaska Native populations and possibly in selected other American Indian populations.
12 Palivizumab prophylaxis is not recommended for prevention of health care-associated RSV disease.
The Departments of Neonatology and Pediatric Pulmonology at The Children’s Hospital suggest that the following groups of patients in your practice be considered as potential benefactors of monthly administered SYNAGISTM immunoprophylaxis from October through March (may need to begin in September or extend through April if our RSV season seems particularly severe and/or prolonged).
1. Infants of any gestational age with BPD requiring medical therapy who are < 2 years old. This category includes infants < 2 years old with a tracheostomy for any reason.
2. Premature infants < 32 completed weeks gestational age and < 1 year old at the start of RSV season.
3. Older premature infants 32 weeks, 0 days through 34 weeks, 6 days gestational age and < 3 months old at the start of RSV season who meet one or more of the following RSV risk criteria:
a. Sibling less than 5 years old at home
b. Daycare exposure
4. Children < 2 years old with hemodynamically significant cyanotic and acyanotic heart disease
Obviously, this latter group comprises the gray zone of risk. We believe that infants who were critically ill in the NICU or PICU and/or who have more than one of the listed RSV risk criteria are most at risk. A twin or triplet who was 34-35 weeks EGA and in good health and who is kept in a home with non-smoking parents and no older siblings is at a very small risk of acquiring RSV and becoming critically ill.
There is a current Synagis® form that we use to send to potential outpatient providers that requires parent signature before faxing to the provider. Please review this form as it may change from year-to-year.
17. Neurodevelopmental Follow Up Program
1. Handwashing
- is required for all parents and health care personnel entering the NICU. Use of soap and water for 2 minutes or alcohol-based hand gels for 20 seconds is satisfactory.
- we expect handwashing to occur prior to and following entering a patient bunker and upon leaving a patient bunker (see definition below)
- No jewelry or sleeves should be worn or extend below the elbow when working in the patient bunker
- The infant
- Any object touching the infant (all electronic leads, blankets, etc.)
- The enclosure containing the infant
- The bedside computer monitor and keyboard
- All electronic monitors and intravenous pumps
- Any other device touching an infant (IVs, central lines, chest tubes, etc.)
- All intravenous lines are to be inserted after sterile skin preparation
- All central lines are to be inserted using complete sterile technique
- Maintenance of and entry into all intravenous lines should be done in a clean fashion after sterile preparation of the access hub
- We strongly recommend that all umbilical lines be removed after 7 days of use
- All infants will have umbilical central lines placed following admission to the NICU if the infant is < 28 weeks gestation or < 1000 grams birth weight
- All sepsis evaluations should include PICC and peripheral blood cultures if a central line is in place
- All infants > 7 days old should also have an Lumbar Puncture performed as part of any sepsis evaluation
- Any infant with neurological symptomatology should have an LP performed as part of a sepsis evaluation (no matter what his or her postnatal age).
Guideline for CSF Reservoir Tap
1. Check patient’s activity, need for suctioning, feeds, etc. with bedside nurse and plan to do the puncture when the patient is not under stress.
2. Position bed flat with patient prone and reservoir side up.
3. Carefully scrub hands and arms to the elbows, put on sterile gloves and wear a mask.
4. Prep skin with betadine swabs and let sit for 3-5 minutes. Use a sterile field around the area of the reservoir.
5. Attach a 25 gauge Butterfly needle to a 5 cc or 10 cc syringe. Insert the needle into the reservoir.
6. Draw off the desired volume of CSF into the syringe SLOWLY (over 10-15 min).
7. Remove the needle and apply pressure with a sterile 2X2 gauze for 1 min.
8. Send CSF sample in three separate tubes for glucose, protein, cell counts and culture at least every other day, and as directed by the Neonatologist.
9. Will need to follow serum Na+ levels and may need to consider intravenous or enteral sodium replacement with long-term reservoir tap.
Guidelines for the Use of Alteplase (Cathflo Activase®) for the
Declotting of Central Lines or to Clear Persistent Central Line
Catheter-Related Bacteremia
Alteplase is approved for the treatment of thrombotic catheter occlusions and has been investigated for the clearance of persistent cathetere-related bacteremia.
Alteplase should not be administered to
Patients with known hypersensitivity to Alteplase or any component of the formulation
Patients with known increased risk of bleeding
Patients with known persistent catheter-related bacteremia unless approved by Pediatric ID.
Alteplase should also be used with caution in
Patients with known active internal bleeding
Patients who have had surgery within the preceeding 48 hours
Patients with thrombocytopenia
Equipment
Alteplase medication (1 mg/mL) in 10 mL syringe drawn up and reconstituted by Pharmacy
Alcohol wipes
Sign or label indication “Do Not Use Catheter – contains Alteplase”
Saline filled 10 mL syringes
Procedure
1) Gather medication and equipment
2) Explain procedure to parents
3) Determine amount of Alteplase required to fill the catheter (no more than 0.5 mL in any catheter). You must avoid giving any volume that may enter the patient.
4) Wash hands
5) Attempt to flush line with normal saline using a 10 mL syringe.
6) Inspect Alteplase syringe for any contaminants or discoloration (should be clear to a slight yellow color).
7) Install Alteplase into catheter (NOT INTO THE PATIENT). Use a push and pull technique with gentle pressure. If resistance is encountered, do not excessive force to place Alteplase in catheter.
8) Place label or sign on catheter to alert others to the presence of an Alteplase-filled catheter.
9) After 30-60 minutes of dwell time, assess catheter function by attempting to aspirate blood (0.5–1 mL for infants < 10 kg). DO NOT INFUSE DRUG INTO PATIENT.
10) If unable to aspirate, allow drug to dwell for another 30-60 minutes of time, then reassess catheter patency
11) If catheter is not patent after 120 minutes of dwell time, you may repeat Alteplase dosing cycle. Maximum dosing for catheter patency is two doses.
Finally, once catheter is patent (you have successfully withdrawn 0.5-1 mL of blood), discard withdrawn blood sample (DO NOT INFUSE DRUG INTO PATIENT) and flush catheter with 0.3 mL of saline followed with an equal amount of heparin solution.